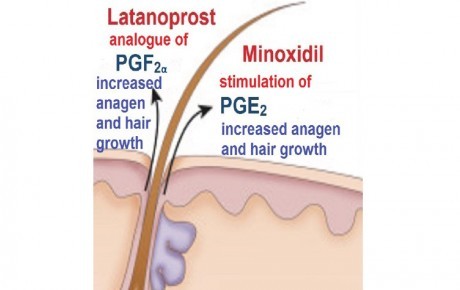
Hair loss is an increasingly common complaint in the field of dermatology, impairing psychosocial well-being and quality of life for affected individuals. The condition is a widespread experience, affecting up to 80% of men [1] and 40% of women [2] in some degree. Patients can be highly motivated to seek diagnosis and treatment [3].
Minoxidil, originally developed for the treatment of hypertension, is the most common topical agent to treat androgenetic alopecia approved by the FDA [4]. Latanoprost, used clinically in the treatment of glaucoma, has been getting a lot of attention lately because of its side effect of inducing growth of lashes and ancillary hairs around the eyelids [5]. A recent double-blind placebo controlled pilot study reported a significant increase in hair density after 24 weeks of topical treatment with 0.1% latanoprost, compared to placebo [6].
Both minoxidil and latanoprost can be made available for topical treatment through compounding. Espumil™ is Fagron’s innovative lipophilic foam base that assures simple application on difficult-to-treat areas, such as hairy skin and the scalp, without dripping. Espumil™ has minimal greasiness, itching, burning, and contact dermatitis due to low concentrations of ethanol and propylene glycol. Foam is generated by foam-activating packaging, without the use of gaseous propellants. Espumil™ has proven to be an ideal and stable vehicle for the application of minoxidil base [7].
[1] Alsantali A, Shapiro J. Androgens and hair loss. Curr Opin Endocrinol Diabetes Obes. 2009; 16:246-253.
[2] Norwood OT. Incidence of female androgenetic alopecia (female pattern alopecia). Dermatol Surg. 2001; 27(1):53-54.
[3] McElwee KJ, Shapiro JS. Promising Therapies for Treating and/or Preventing Androgenic Alopecia. Skin Therapy Lett. 2012 Jun; 17(6):1-4.
[4] Shin HS, Won CH, Lee SH, Kwon OS, Kim KH et al. Efficacy of 5% Minoxidil versus Combined 5% Minoxidil and 0.01% Tretinoin for Male Pattern Hair Loss. Am J Clin Dermatol. 2007;8(5):285-290.
[5] Johnstone MA, Albert DM. Prostaglandin-induced hair growth. Surv Ophthalmol. 2002 Aug;47 Suppl 1:S185-202.
[6] Blume-Peytavi U, Lönnfors S, Hillmann K, Garcia Bartels N. A randomized double-blind placebo controlled pilot study to assess the efficacy of a 24-week topical treatment by latanoprost 0.1% on hair
growth and pigmentation in healthy volunteers with androgenetic alopecia. J Am Acad Dermatol. 2012 May; 66(5):794-800.
[7] Geiger CM, Sorenson B, Whaley PA. Stability of Minoxidil in Espumil foam base. IJPC. 2013;17(2):165-167. [link]